SARS-CoV-2 recombinant Spike (SCoV2-rS) glycoprotein trimeric ectodomain from mammalian HEK293 cells, frozen
Description: SARS-CoV-2 recombinant Spike (SCoV2-rS) glycoprotein ectodomain expressed as secreted trimeric protein in mammalian (human) HEK293 cells. UniProtKB sequence accession number: P0DTC2 (SPIKE_SARS2); expressed ectodomain includes amino acids (aa) 1-1208. Whole expression construct includes: full-length SCoV2-S ectodomain (aa 1 – 1208) w/o transmembrane and cytoplasmic aa, furin cleavage site “RRAR” mutated to “GSAS”, C-terminal GCN4 trimerization motif fused to protein sequence, then follows thrombin cleavage site, Strep-tag II and His6-tag. Two mutations (K986P and V987P) were introduced into SCoV2-S sequence to stabilize the trimer in the pre-fusion conformation. SCoV2-rS glycoprotein was purified from culture medium under native (non-denaturing) conditions using convenient chromatographic techniques. Then it was dialyzed against PBS, adjusted to 1.0 mg/ml concentration, filter-sterilized, aliquoted and frozen for storage.
Form: Frozen liquid in PBS (pH 7.4).
Storage: The product is shipped with dry ice (shipment cost to Europe: 160 €). Store at -80°C for the long term. After thawing may be stored for short-term at 4°C (stable at least 2 weeks at this temperature). Avoid repeated freeze/thaw cycles.
Concentration: 1.0 mg/ml.
Purity: >90 % as determined by SDS-PAGE.
Image: SDS-PAGE showing SCoV2-rS product 20-S2S-THs-F at approximately 160 kDa (1, 3 and 5 μg/lane).
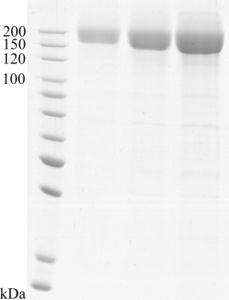
Molecular weight (MW): Predicted MW of SCoV2-rS is 142.11 kDa. Expression in mammalian cells ensures authentic post-translational modifications including glycosylation. Glycosylation increases MW, therefore SCoV2-rS is detected as intact protein at ~160 kDa (from CHO cells) or at 160-200 kDa (from HEK293 cells), respectively. The observed differences in glycosylation extent did not result in significant differences in anti-SARS-CoV-2 IgG ELISA assay.
Applications (tested): ELISA, Western blot, SDS-PAGE.
Quality: According to the expressed construct, purified SCoV2-rS protein should be trimer in the stabilized prefusion conformation. Full-length trimeric protein enables detection of more antibodies against SCoV2-S, than shorter protein constructs (e.g., RBD or S1). This product 20-S2S-THs-F showed similar results in ELISA with COVID-19 positive and negative patient sera, as analogous protein construct derived from CHO cells (see product 20-S2S-TCg-G information), but a lesser number of individual sera was tested. SCoV2-rS from HEK293 cells was not selected for further applications in broader population studies, because analogous product from CHO cells seems more convenient (due to more uniform glycosylation and higher yields).
Unit/Price:
| Size | Catalogue No. | Price | |
| 50 μg | 20-S2S-THs-F-L | 375 € | Inquiry / Order product |
| 100 μg | 20-S2S-THs-F-C | 675 € | |
| 500 μg | 20-S2S-THs-F-D | 2500 € |