SARS-CoV-2 nucleoprotein C-terminal domain from S. cerevisiae, frozen
Description: SARS-CoV-2 recombinant Nucleoprotein (SCoV2-rN) with deletion of N-terminal domain (SCoV2-ΔN-rN) expressed in yeast Saccharomyces cerevisiae. The construct contains amino acids 121-419 of the Nucleoprotein provided in UniProtKB sequence accession number: P0DTC9 (NCAP_SARS2), expressed with C-terminal His6-tag. It was purified from yeast lysates under native (non-denaturing) conditions using convenient chromatographic techniques. Then it was dialyzed against PBS, adjusted to 1.0 mg/ml concentration, filter-sterilized, aliquoted and frozen for storage.
Form: Frozen liquid in PBS (pH 7.4) with 500 mM NaCl.
Safety: Product 21-S2delNN-ScB-F contains only yeast expressed proteins that are not infectious or hazard. Contains no plasma or serum of animal origin. No safety precautions are required.
Storage: The product is shipped with dry ice (shipment cost to Europe: 160 €). Store at -80°C for the long term. After thawing may be stored for short-term at 4°C (stable at least 1 week at this temperature). Avoid repeated freeze/thaw cycles.
Concentration: 1.0 mg/ml.
Image: SDS-PAGE and Western blots showing SCoV2-ΔN-rN product 21-S2delNN-ScB-F (ΔN lanes) at approximately 40 kDa (3 μg/lane) with two bands of truncated forms that react with monoclonal antibodies. For comparison, a full-length SCoV2-rN product 20-S2N-ScB-F (lanes N03) and different amounts of BSA standard (BSA lanes) are shown. “M” indicates Mw marker “PageRuler™ Prestained Protein Ladder“ (ThermoFisher, Cat. No. #26617).
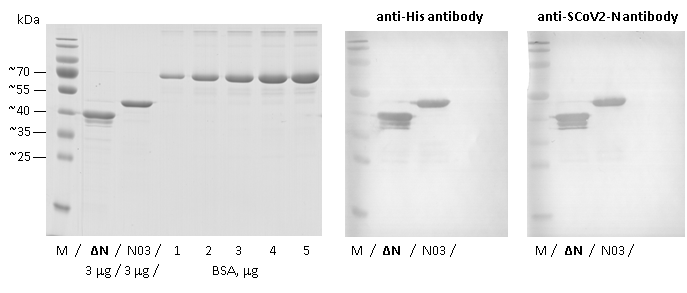
Purity: >90% pure SCoV2-ΔN-rN protein including two truncated forms, as determined by SDS-PAGE.
Applications (tested): ELISA, other immunoassays, Western blot, SDS-PAGE.
Note: Such truncated SCoV2-rN construct without 121 aa of N-terminal domain has been reported to improve specificity in serological assays compared to the full-length N protein (Ref. 1-3).
Unit/Price:
| Size | Catalogue No. | Price | |
| 100 μg | 21-S2delNN-ScB-F-C | 200 € | Inquiry / Order product |
| 500 μg (5×100 μg) | 21-S2delNN-ScB-F-D | 666 € | |
| 1 mg (10×100 μg) | 21-S2delNN-ScB-F-M | 1000€ |
References:
- Yamaoka Y, Jeremiah SS, Miyakawa K, Saji R, Nishii M, Takeuchi I, Ryo A: Whole Nucleocapsid Protein of Severe Acute Respiratory Syndrome Coronavirus 2 May Cause False-Positive Results in Serological Assays. Clin Infect Dis. 2021 Apr 8; 72(7):1291-1292. doi: 10.1093/cid/ciaa637.
- Mutantu PN, Ngwe Tun MM, Nabeshima T, Yu F, Mukadi PK, Tanaka T, Tashiro M, Fujita A, Kanie N, Oshiro R, Takazono T, Imamura Y, Hirayama T, Moi ML, Inoue S, Izumikawa K, Yasuda J, Morita K: Development and Evaluation of Quantitative Immunoglobulin G Enzyme-Linked Immunosorbent Assay for the Diagnosis of Coronavirus Disease 2019 Using Truncated Recombinant Nucleocapsid Protein as Assay Antigen. Int J Environ Res Public Health. 2021 Sep 13; 18(18):9630. doi: 10.3390/ijerph18189630.
- Yue L., Cao H., Xie T., Long R., Li H., Yang T., Yan M., Xie Z. N-terminally truncated nucleocapsid protein of SARS-CoV-2 as a better serological marker than whole nucleocapsid protein in evaluating the immunogenicity of inactivated SARS-CoV-2. Med. Virol. 2021; 93:1732–1738. doi: 10.1002/jmv.26541.