SARS-CoV-2 recombinant Spike (SCoV2-rS) glycoprotein trimeric ectodomain from mammalian CHO cells, frozen
Description: SARS-CoV-2 recombinant Spike (SCoV2-rS) glycoprotein ectodomain expressed as secreted trimeric protein in mammalian (hamster) CHO cells. UniProtKB sequence accession number: P0DTC2 (SPIKE_SARS2); expressed ectodomain includes amino acids (aa) 1-1208. Whole expression construct includes: full-length SCoV2-S ectodomain (aa 1 – 1208) w/o transmembrane and cytoplasmic aa, furin cleavage site “RRAR” mutated to “GSAS”, C-terminal GCN4 trimerization motif fused to protein sequence, then follows thrombin cleavage site, Strep-tag II and His6-tag. Two mutations (K986P and V987P) were introduced into SCoV2-S sequence to stabilize the trimer in the pre-fusion conformation. SCoV2-rS glycoprotein was purified from culture medium under native (non-denaturing) conditions using convenient chromatographic techniques. Then it was dialyzed against PBS, adjusted to 1.0 mg/ml concentration, filter-sterilized, aliquoted and frozen for storage.
Form: Frozen liquid in PBS (pH 7.4).
Storage: The product is shipped with dry ice (shipment cost to Europe: 160 €). Store at -80°C for the long term. After thawing may be stored for short-term at 4°C (stable at least 2 weeks at this temperature). Avoid repeated freeze/thaw cycles.
Concentration: 1.0 mg/ml.
Purity: >90 % as determined by SDS-PAGE.
Image: SDS-PAGE showing SCoV2-rS product 20-S2S-TCg-F at approximately 160 kDa (1, 3 and 5 μg/lane).
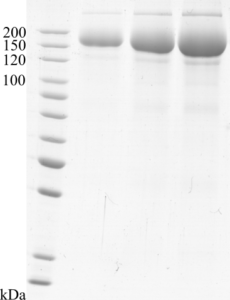
Applications (tested): ELISA, other immunoassays, Total Internal Reflection Ellipsometry (TIRE), Electron microscopy, Western blot, SDS-PAGE.
Quality: According to the expressed construct, purified SCoV2-rS protein should be trimer in the stabilized prefusion conformation. Full-length trimeric protein enables detection of more antibodies against SCoV2-S, than shorter protein constructs (e.g., RBD or S1). It provides a tool to develop highly sensitive classical immunoassays, as well as design novel physico-chemical methods for monitoring interactions of SCoV2-S antigens with specific antibodies (see References). This product 20-S2S-TCg-F showed similar results in ELISA with COVID-19 positive and negative patient sera, as analogous protein construct formulated in glycerol stock (see product 20-S2S-TCg-G information), but a lesser number of individual sera was tested. Besides, it was successfully used for the development of several electrochemical and optical methods including the highly sensitive total internal reflection ellipsometry method for the evaluation of interaction kinetics between SCoV2-S and specific antibodies in human serum samples (Ref. 1).
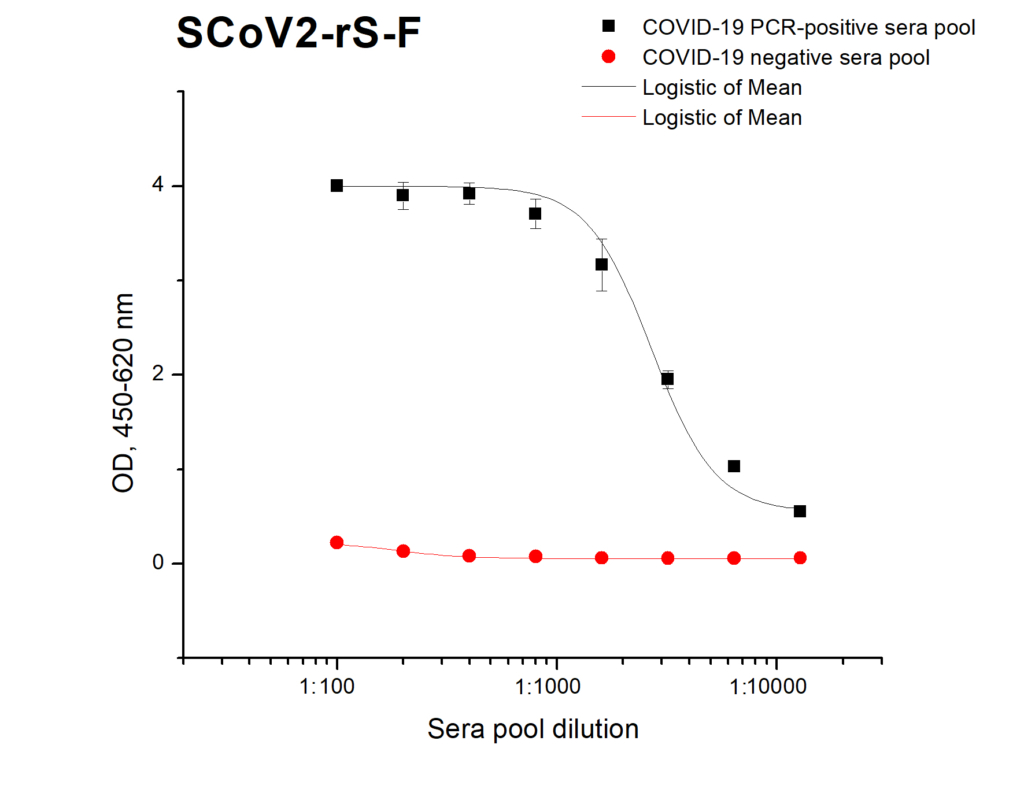
Reactivity of SCoV2-rS glycoprotein (current batch of product 20-S2S-TCg-F) with specific IgG in ELISA using pools of Covid-19 positive and negative sera:
Interaction kinetics measured between specific polyclonal human antibodies against SARS-CoV2 S protein (pAb-SCoV2-S) produced after vaccination with the Vaxzevria vaccine and SCoV2-rS glycoprotein (product 20-S2S-TCg-F) by total internal reflection ellipsometry (TIRE) method: association and dissociation rate constants, the equilibrium association (KA) and dissociation (KD) constants were calculated using a two-step irreversible binding mathematical model (Ref. 1). The results showed high-affinity binding of vaccine-induced pAb-SCoV2-S antibodies from human blood serum to the SCoV2-rS glycoprotein (Ref. 1). This enables monitoring of the humoral immune response by a highly sensitive, label-free, and real-time TIRE method after vaccination with COVID-19 vaccines.
Unit/Price:
| Size | Catalogue No. | Price | |
| 50 μg | 20-S2S-TCg-F-L | 325 € | Inquiry / Order product |
| 100 μg | 20-S2S-TCg-F-C | 500 € | |
| 500 μg | 20-S2S-TCg-F-D | 1875 € | |
| 1 mg | 20-S2S-TCg-F-M | 3125 € |
References on the use of this product (20-S2S-TCg-F):
1. Plikusiene I, Maciulis V, Juciute S, Maciuleviciene R, Balevicius S, Ramanavicius A, Ramanaviciene A: Investigation and Comparison of Specific Antibodies’ Affinity Interaction with SARS-CoV-2 Wild-Type, B.1.1.7, and B.1.351 Spike Protein by Total Internal Reflection Ellipsometry. Biosensors (Basel). 2022 May 18; 12(5):351. doi: 10.3390/bios12050351. Full text.
2. Ratautaite V, Boguzaite R, Brazys E, Ramanaviciene A, Ciplys E, Juozapaitis M, Slibinskas R, Bechelany M, Ramanavicius A: Molecularly imprinted polypyrrole based sensor for the detection of SARS-CoV-2 spike glycoprotein. Electrochim Acta. 2022 Jan 20; 403:139581. doi: 10.1016/j.electacta.2021.139581.
3. Drobysh M, Liustrovaite V, Baradoke A, Rucinskiene A, Ramanaviciene A, Ratautaite V, Viter R, Chen CF, Plikusiene I, Samukaite-Bubniene U, Slibinskas R, Ciplys E, Simanavicius M, Zvirbliene A, Kucinskaite-Kodze I, Ramanavicius A: Electrochemical Determination of Interaction between SARS-CoV-2 Spike Protein and Specific Antibodies. Int J Mol Sci. 2022 Jun 17; 23(12):6768. doi: 10.3390/ijms23126768.
4. Drobysh M, Liustrovaite V, Baradoke A, Viter R, Chen CF, Ramanavicius A, Ramanaviciene A: Determination of rSpike Protein by Specific Antibodies with Screen-Printed Carbon Electrode Modified by Electrodeposited Gold Nanostructures. Biosensors (Basel). 2022 Aug 3; 12(8):593. doi: 10.3390/bios12080593.
5. Zukauskas S, Rucinskiene A, Ratautaite V, Ramanaviciene A, Pilvenyte G, Bechelany M, Ramanavicius A: Electrochemical Biosensor for the Determination of Specific Antibodies against SARS-CoV-2 Spike Protein. Int J Mol Sci. 2022 Dec 31; 24(1):718. doi: 10.3390/ijms24010718.
6. Liustrovaite V.; Drobysh, M.; Rucinskiene, A.; Baradoke, A.; Ramanaviciene, A.; Plikusiene, I.; Samukaite-Bubniene, U.; Viter, R.; Chen, C.-F.; Ramanavicius, A. Towards an Electrochemical Immunosensor for the Detection of Antibodies against SARS-CoV-2 Spike Protein. J. Electrochem. Soc. 2022, 169, 037523. DOI: 10.1149/1945-7111/ac5d91.
7. Drobysh M, Ramanavicius A, Baradoke A: Polyaniline-based electrochemical immunosensor for the determination of antibodies against SARS-CoV-2 spike protein. Sci Total Environ. 2023 Mar 1; 862:160700. doi: 10.1016/j.scitotenv.2022.160700.